
Diboron-Promoted Reduction of Ni(II) Salts: Precatalyst Activation Studies Relevant to Ni-Catalyzed Borylation Reactions | Organometallics

Dual electronic effects achieving a high-performance Ni(II) pincer catalyst for CO2 photoreduction in a noble-metal-free system | PNAS

Inorganics | Free Full-Text | Nickel(II) N-Heterocyclic Carbene Complex for the Hydrogenation of 2-Acetylpyridine under Mild Conditions

Spectroelectrochemical, photochemical and theoretical study of octaazamacrocyclic nickel(II) complexes exhibiting unusual solvent-dependent deprotonation of methylene group - ScienceDirect

Catalysts | Free Full-Text | New Ni(II)-Ni(II) Dinuclear Complex, a Resting State of the (α-diimine)NiBr2/AlMe3 Catalyst System for Ethylene Polymerization

PDF) Nickel(II) complex incorporating methylene bridged tetradentatedicarbene ligand as an efficient catalyst toward C-C and C-N bondformation reactions | Nirmala Muthukumaran - Academia.edu
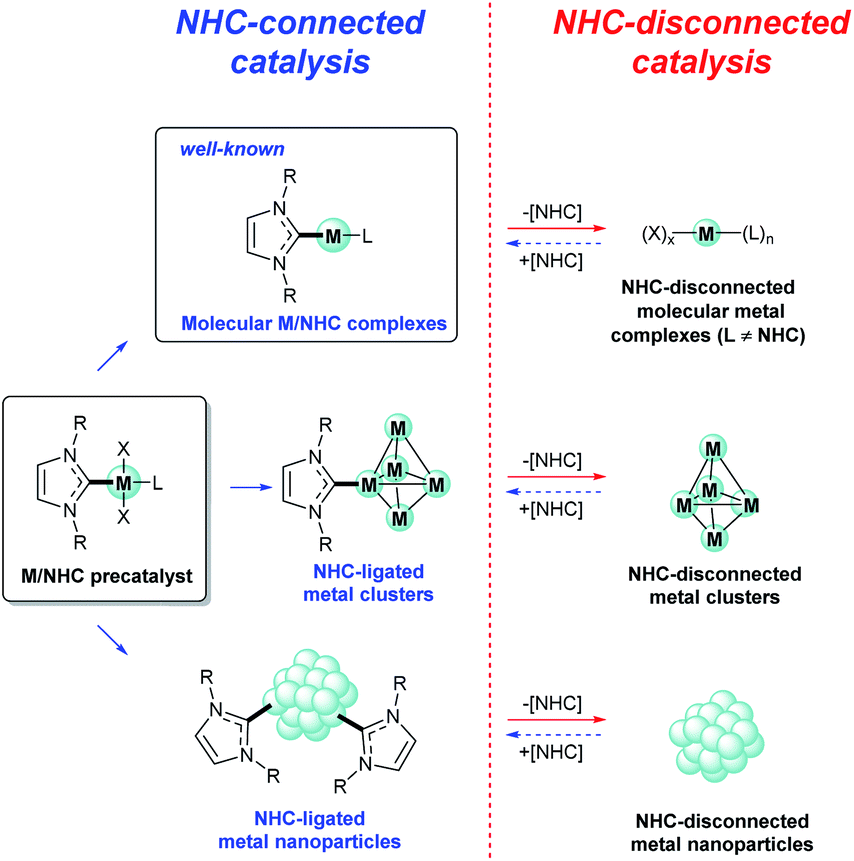
The key role of R–NHC coupling (R = C, H, heteroatom) and M–NHC bond cleavage in the evolution of M/NHC complexes and formation of catalytically activ ... - Chemical Science (RSC Publishing)
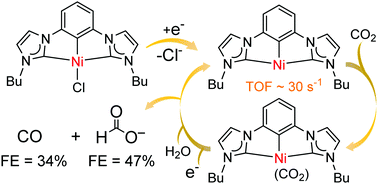
Electrocatalytic reduction of CO2 with CCC-NHC pincer nickel complexes - Chemical Communications (RSC Publishing)

Synthesis and structural characterization of nickel(II) complexes of 20-membered macrocyclic rings bearing chelating bis(N-heterocyclic carbene) ligands - ScienceDirect

An Unsaturated Nickel(0) NHC Catalyst: Facile Preparation and Structure of Ni(0)(NHC)2, Featuring a Reduction Process from Ni(II)(NHC)(acac)2 | Organometallics

Molecules | Free Full-Text | Recent Advances in Catalysis Involving Bidentate N-Heterocyclic Carbene Ligands

Well-defined nickel(II) tetrazole-saccharinate complex as homogeneous catalyst on the reduction of aldehydes: scope and reaction mechanism

Nickel(II) N‐Heterocyclic Carbene Complexes: Versatile Catalysts for C–C, C–S and C–N Coupling Reactions - Junquera - 2017 - European Journal of Inorganic Chemistry - Wiley Online Library

Synthesis and Characterization of Bidentate NHC-CAryl Nickel(II) Complexes: Isocyanide Insertion To Form NHC-η2-iminoacyl Complexes | Organometallics

An Unsaturated Nickel(0) NHC Catalyst: Facile Preparation and Structure of Ni(0)(NHC)2, Featuring a Reduction Process from Ni(II)(NHC)(acac)2 | Organometallics

3d-d Excited States of Ni(II) Complexes Relevant to Photoredox Catalysis: Spectroscopic Identification and Mechanistic Implications | Journal of the American Chemical Society
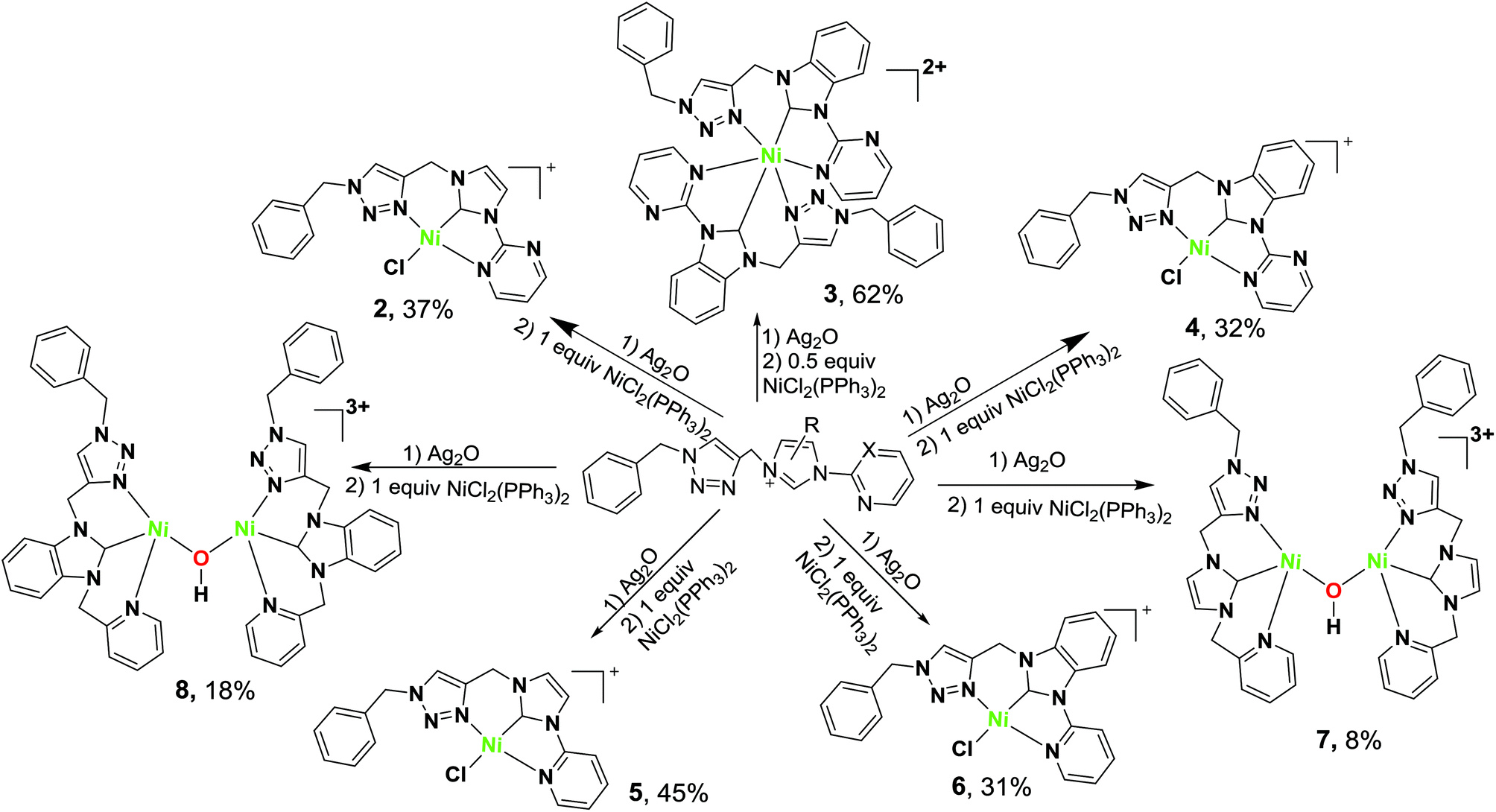
Unsymmetrical NCN-pincer mononuclear and dinuclear nickel( ii ) complexes of N-heterocyclic carbene (NHC): synthesis, structure and catalysis for Suzu ... - Dalton Transactions (RSC Publishing) DOI:10.1039/C6DT03944H

Multidentate N‐Heterocyclic Carbene Complexes of Nickel and Palladium: Structural Analysis and Catalytic Application in Mizoroki–Heck Coupling Reaction - Liu - 2020 - European Journal of Inorganic Chemistry - Wiley Online Library
![Well‐Defined Allylnickel Chloride/N‐Heterocyclic Carbene [(NHC)Ni(allyl)Cl] Complexes as Highly Active Precatalysts for CN and CS Cross‐Coupling Reactions - Iglesias - 2010 - Advanced Synthesis & Catalysis - Wiley Online Library Well‐Defined Allylnickel Chloride/N‐Heterocyclic Carbene [(NHC)Ni(allyl)Cl] Complexes as Highly Active Precatalysts for CN and CS Cross‐Coupling Reactions - Iglesias - 2010 - Advanced Synthesis & Catalysis - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/eb9616bd-721b-475e-bfa1-7e91c16670c5/msch001.gif)
Well‐Defined Allylnickel Chloride/N‐Heterocyclic Carbene [(NHC)Ni(allyl)Cl] Complexes as Highly Active Precatalysts for CN and CS Cross‐Coupling Reactions - Iglesias - 2010 - Advanced Synthesis & Catalysis - Wiley Online Library

Well-defined nickel(II) tetrazole-saccharinate complex as homogeneous catalyst on the reduction of aldehydes: scope and reaction mechanism
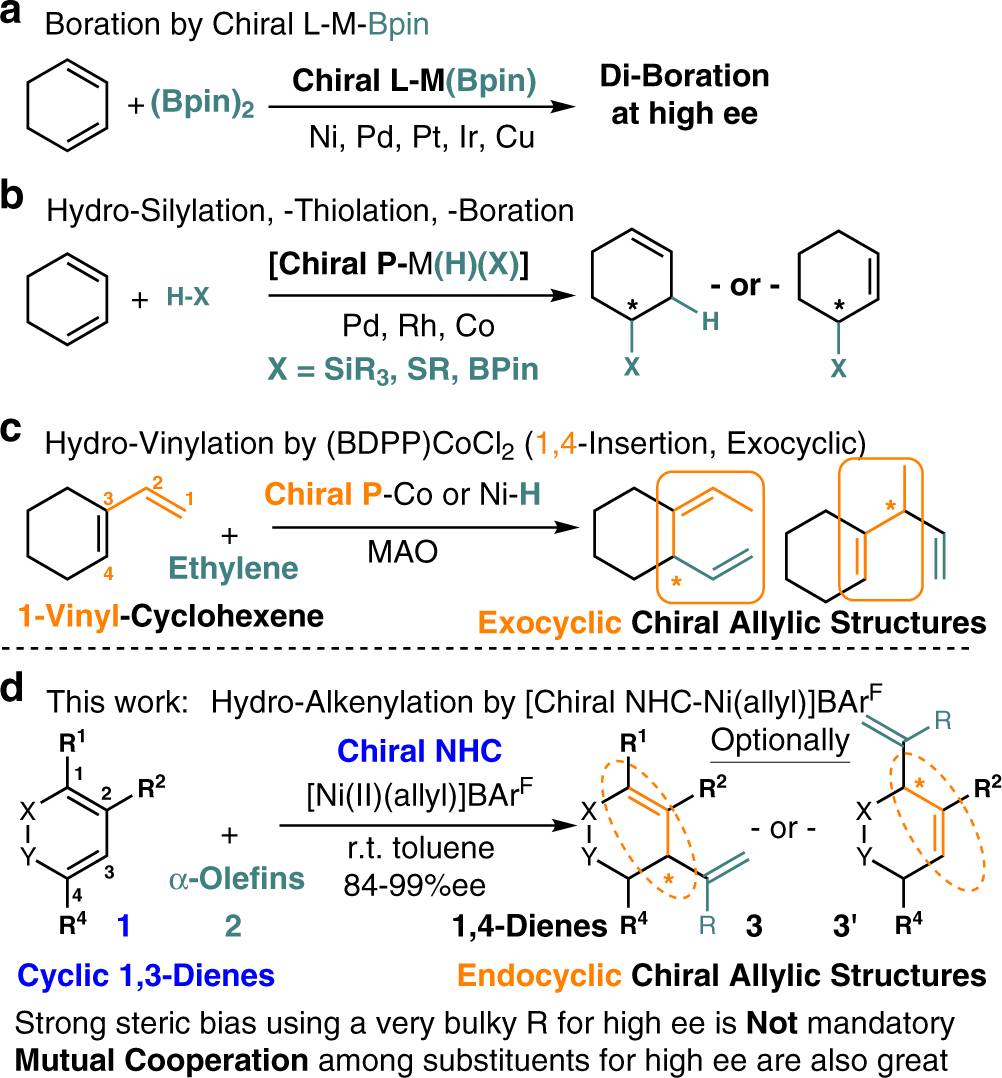
NHC-Ni catalyzed enantioselective synthesis of 1,4-dienes by cross-hydroalkenylation of cyclic 1,3-dienes and heterosubstituted terminal olefins | Nature Communications
![Ni(NHC)2] as a Scaffold for Structurally Characterized trans [H−Ni−PR2] and trans [R2P−Ni−PR2] Complexes - Sabater - 2021 - Chemistry – A European Journal - Wiley Online Library Ni(NHC)2] as a Scaffold for Structurally Characterized trans [H−Ni−PR2] and trans [R2P−Ni−PR2] Complexes - Sabater - 2021 - Chemistry – A European Journal - Wiley Online Library](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/f9899e7f-8771-4ad0-b47c-06e193f2a547/chem202101484-toc-0001-m.jpg)
Ni(NHC)2] as a Scaffold for Structurally Characterized trans [H−Ni−PR2] and trans [R2P−Ni−PR2] Complexes - Sabater - 2021 - Chemistry – A European Journal - Wiley Online Library
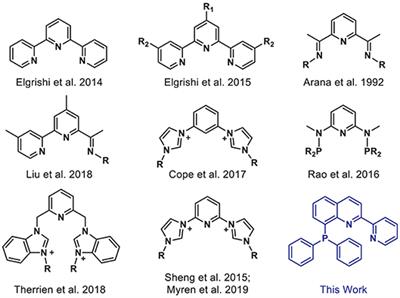


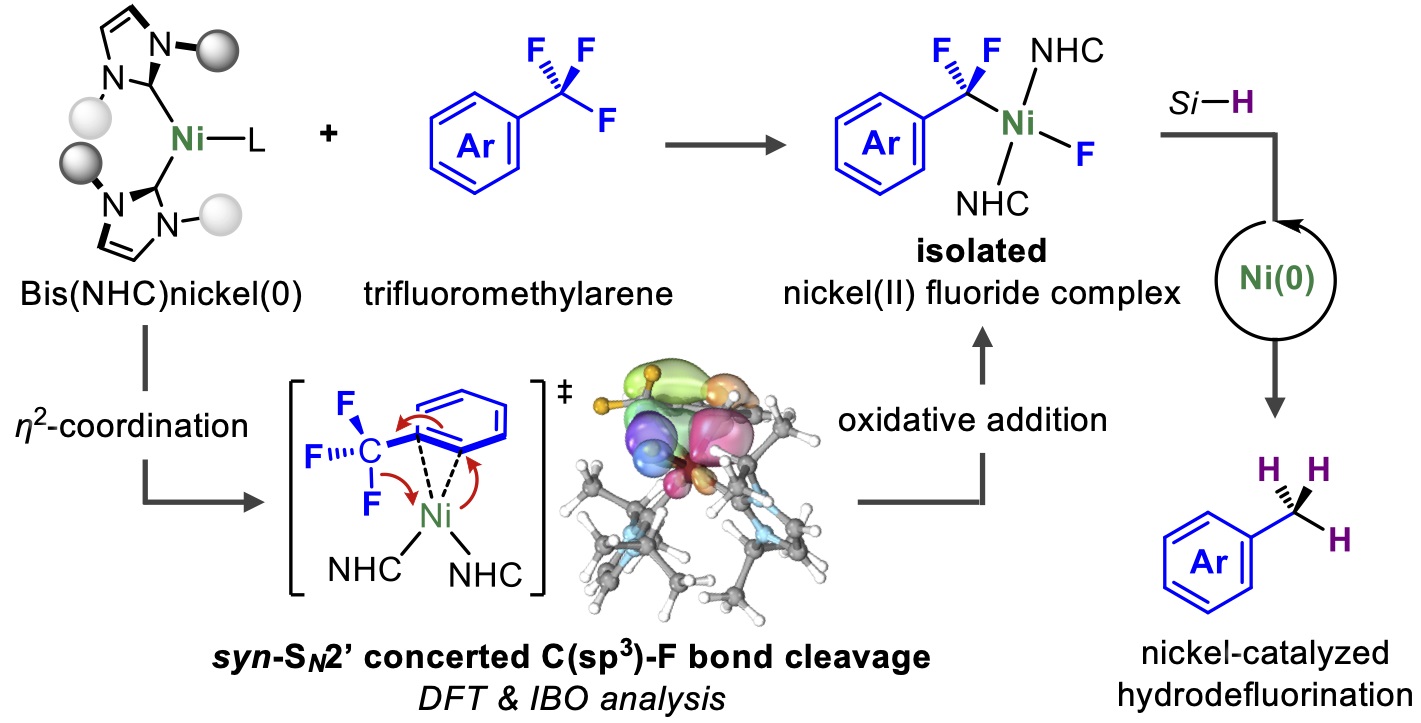

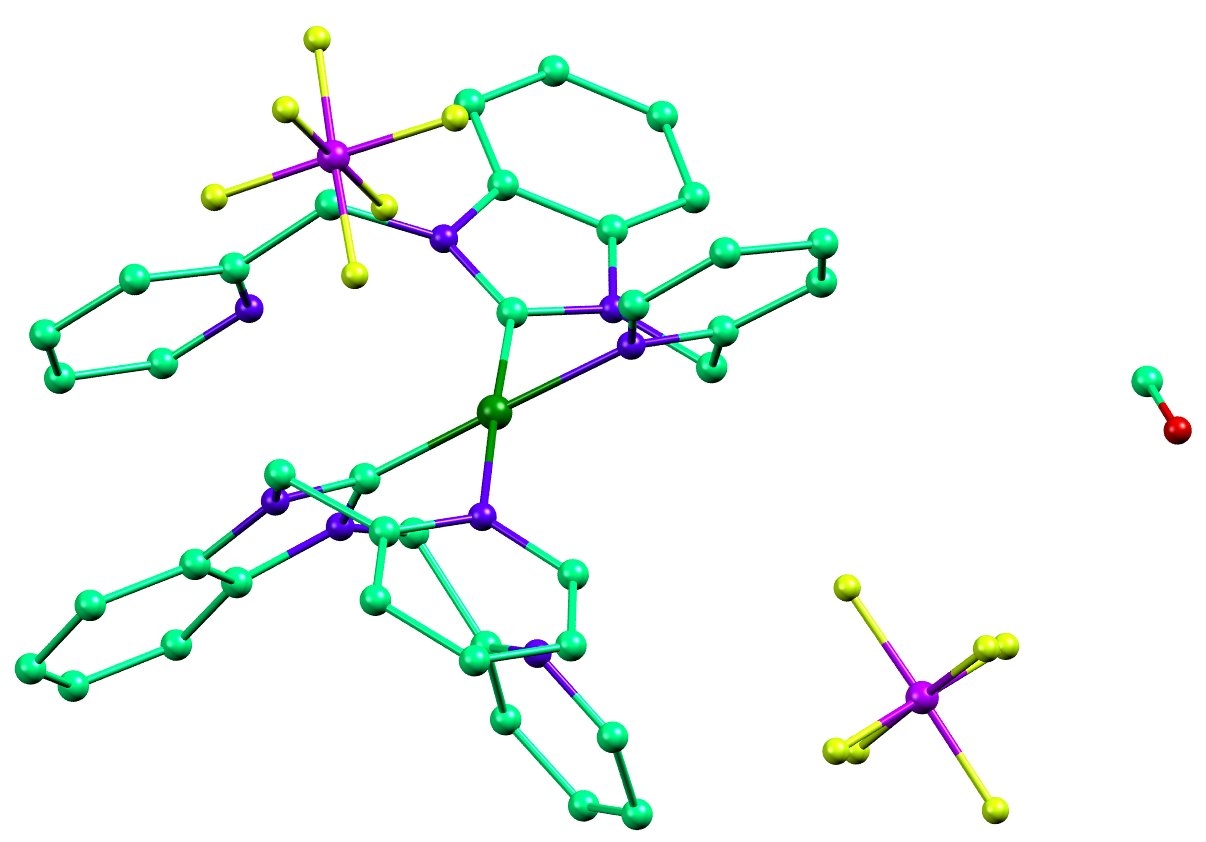

![NHC-Ni(II)-catalyzed cyclopropene-isocyanide [5 + 1] benzannulation | Nature Communications NHC-Ni(II)-catalyzed cyclopropene-isocyanide [5 + 1] benzannulation | Nature Communications](https://media.springernature.com/m685/springer-static/image/art%3A10.1038%2Fs41467-022-31896-y/MediaObjects/41467_2022_31896_Fig1_HTML.png)